Chemistry - Atomic Structure Question with Solution | TestHub
ChemistryAtomic StructureSchrödinger's EquationMedium2 minPYQ_2021
ChemistryMediummatching list
The plots of radial distribution functions for various orbitals of hydrogen atom against 'are given below.
The correct plot for orbital is :
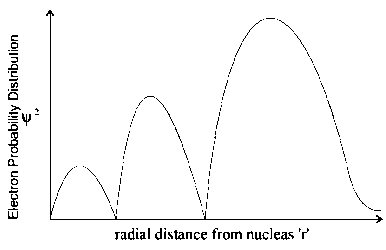
Options:
Answer:
C
Solution:
Calculate the number of radial nodes using the
Number of radial nodes
Radial nodes= number of cuts on x-axis ( r).
The curve should start from zero. It can be positive or negative.
Size of the curve increases after touching the x- axis every time .
By keeping all this, we get the following curve:

Therefor corresponding graph is (C)
Hence, answer is
Stream:JEESubject:ChemistryTopic:Atomic StructureSubtopic:Schrödinger's Equation
⏱ 2mℹ️ Source: PYQ_2021
Doubts & Discussion
Loading discussions...