Chemistry - Mole Concept Question with Solution | TestHub
ChemistryMole ConceptMiscellaneous/MixedHard2 minPYQ_2019
ChemistryHardassertion reason
Atandatmospheric pressure,of a hydrocarbon requiredoffor complete combustion, andofis formed. The formula of the hydrocarbon is:
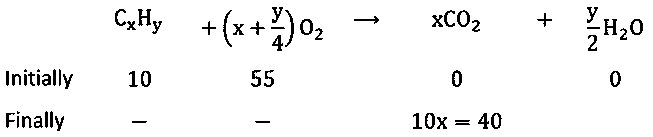
Options:
Answer:
C
Solution:
Let hydrocarbon is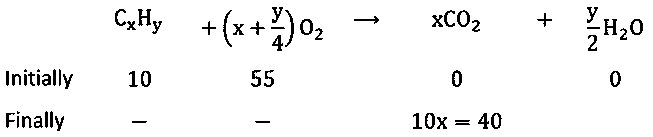
;and
Put the value of
then
Hence hydrocarbon is
Stream:JEESubject:ChemistryTopic:Mole ConceptSubtopic:Miscellaneous/Mixed
⏱ 2mℹ️ Source: PYQ_2019
Doubts & Discussion
Loading discussions...