Chemistry - Liquid Solution Question with Solution | TestHub
ChemistryLiquid SolutionHenry's LawHard2 minPYQ_2019
ChemistryHardsingle choice
For the solution of the gasesandin water atthe Henry's law constantsareandrespectively. The correct plot for the given data is:
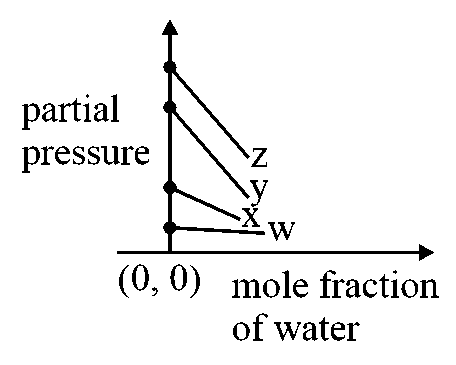
Options:
Answer:
A
Solution:
Where, is the partial pressure of gas.
is the Henry’s constant
is the mole fraction of gas
On comparing this with the equation of straight line, , the intercept, . Thus, the gas having more value of Henry's law constant will have more value of intercept on the y-axis.
Stream:JEESubject:ChemistryTopic:Liquid SolutionSubtopic:Henry's Law
⏱ 2mℹ️ Source: PYQ_2019
Doubts & Discussion
Loading discussions...