Chemistry - Electrochemistry Question with Solution | TestHub
The number of correct statements from the following is ___________
(A) Conductivity always decreases with decrease in concentration for both strong and weak electrolytes.
(B) The number of ions per unit volume that carry current in a solution increases on dilution.
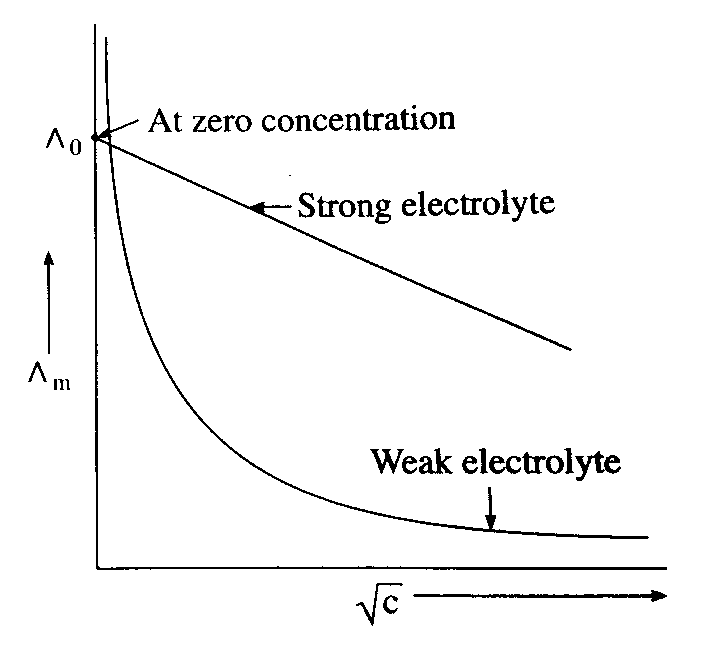
(C) Molar conductivity increases with decrease in concentration.
(D) The variation in molar conductivity is different for strong and weak electrolytes.
(E) For weak electrolytes, the change in molar conductivity with dilution is due to decrease in degree of dissociation.
Answer:
Solution:
Conductivity always decreases with the decrease in concentration both, for weak and strong electrolytes.
This can be explained by the fact that the number of ions per unit volume that carry the current in a solution decreases on dilution.
Molar conductivity of a strong and weak electrolyte increases with dilution. On dilution as volume of solution increases. Thus, on dilution, ions get more apart and mobility of ions increases which leads to increase in molar conductivity of the solution.
Except and , all statements are correct.