Chemistry - Electrochemistry Question with Solution | TestHub
ChemistryElectrochemistryGalvanic cells/Nernst Equation/Concentration CellsMedium2 minPYQ_2020
ChemistryMediumstatement
Given that the standard potentialsofandareandrespectively, theofis:
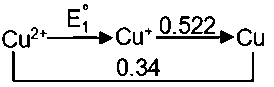
Options:
Answer:
B
Solution:
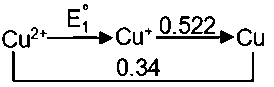
Stream:JEESubject:ChemistryTopic:ElectrochemistrySubtopic:Galvanic cells/Nernst Equation/Concentration Cells
⏱ 2mℹ️ Source: PYQ_2020
Doubts & Discussion
Loading discussions...