Chemistry - Atomic Structure Question with Solution | TestHub
ChemistryAtomic StructureBohr's Atomic ModelMedium2 minPYQ_2023
ChemistryMediummatching list
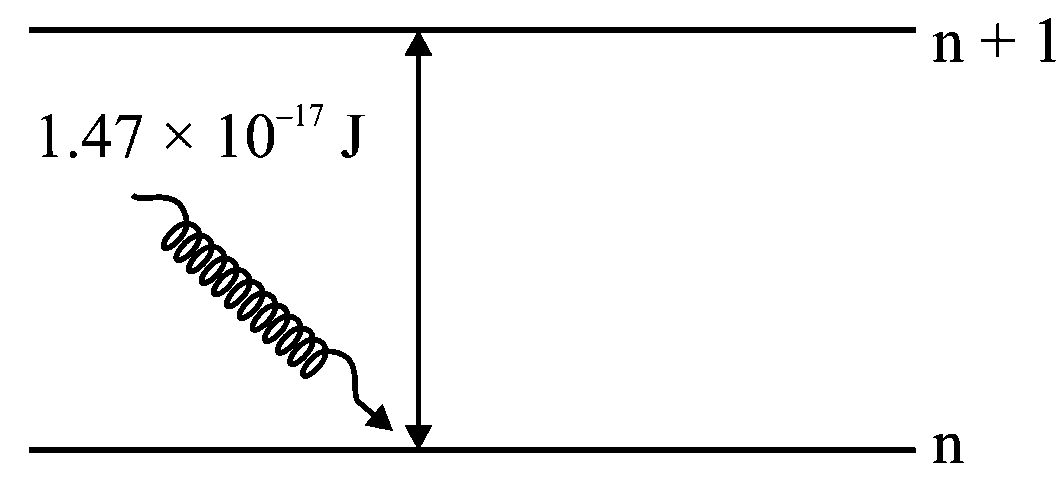
The electron in the orbit of is excited to orbit using the radiation of energy (as shown in the diagram). The value of is ___________
Given :

Answer:
1
Solution:
The difference energy between two energy levels is
Hence, the transition is taking place between , and it comes under Lymann series.
If we take
Stream:JEESubject:ChemistryTopic:Atomic StructureSubtopic:Bohr's Atomic Model
⏱ 2mℹ️ Source: PYQ_2023
Doubts & Discussion
Loading discussions...