Chemistry - Thermochemistry Question with Solution | TestHub
From the following data at
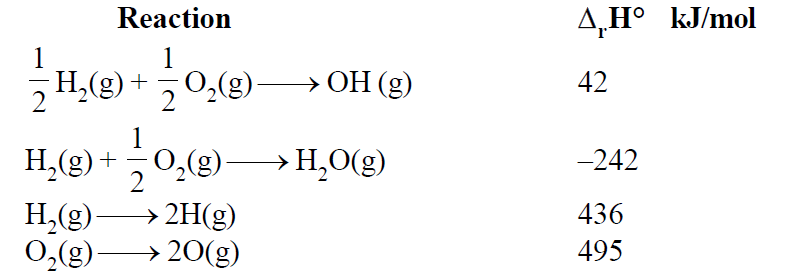
Which of the following statement(s) is/are correct:
Statement (a) : for the reaction is
Statement (b) : for the reaction is
Statement (c) : Enthalpy of formation of is
Statement (d) : Enthalpy of formation of is
Options:
Answer:
Solution:
Correct statements are a and b. The correct option is therefore not A. I will provide a detailed explanation of each statement.
Statement (a): for the reaction is
To calculate the enthalpy change for this reaction, we can use Hess's Law and the given data. We need to manipulate the given reactions to arrive at the desired reaction.
1.
2.
3.
We need to reverse reaction (1) and halve reaction (3):
Adding this to reaction (2):
Hence CORRECT
Statement (b): for the reaction is
We can obtain this by using the following reactions:
1.
2.
3.
We need to reverse reaction (1) and halve reactions (2) and (3):
Adding these together:
The provided value of is incorrect.
Statement (c): Enthalpy of formation of is
The enthalpy of formation of is half the enthalpy change for the reaction , which is . Therefore, the enthalpy of formation of is . The given value of is incorrect.
Statement (d): Enthalpy of formation of is
The enthalpy of formation of is given directly as for the reaction . This statement is correct.