Chemistry - Redox Reactions Question with Solution | TestHub
ChemistryRedox ReactionsBalancing of Redox ReactionsMedium2 minPYQ_2019
ChemistryMediummatching list
An example of a disproportionation reaction is
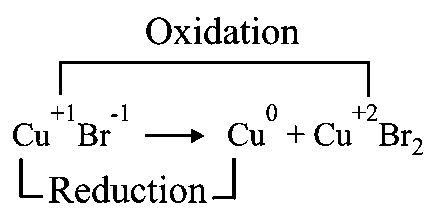
Options:
Answer:
C
Solution:
Disproportionation reaction: In this type of reaction, same elemental species gets oxidised as well as reduced.
not disproportionate,
not disproportionate,
not disproportionate

-It is disproportionate.
Stream:JEESubject:ChemistryTopic:Redox ReactionsSubtopic:Balancing of Redox Reactions
⏱ 2mℹ️ Source: PYQ_2019
Doubts & Discussion
Loading discussions...