Chemistry - Metallurgy Question with Solution | TestHub
ChemistryMetallurgyEllingham diagram & Reduction methods, Refining of metalMedium2 minPYQ_2018
ChemistryMediumnumerical
Galena (an ore) is partially oxidized by passing air through it at high temperature. After some time, the passage of air is stopped, but the heating is continued in a closed furnace such that the contents undergo self-reduction. The weight (in) ofproduced perofconsumed is ____. (Atomic weights in)
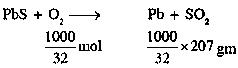
Answer:
6.47
Solution:
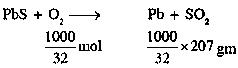
Mass of
Stream:JEE_ADVSubject:ChemistryTopic:MetallurgySubtopic:Ellingham diagram & Reduction methods, Refining of metal
⏱ 2mℹ️ Source: PYQ_2018
Doubts & Discussion
Loading discussions...