Chemistry - Ionic Equilibrium Question with Solution | TestHub
ChemistryIonic EquilibriumSolubility ProductMedium2 minPYQ_2019
ChemistryMediumstatement
Ifofis, the molar solubility ofinis:

Options:
Answer:
D
Solution:
(i) 
(ii) 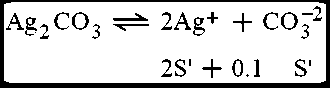
or
Stream:JEESubject:ChemistryTopic:Ionic EquilibriumSubtopic:Solubility Product
⏱ 2mℹ️ Source: PYQ_2019
Doubts & Discussion
Loading discussions...