Chemistry - Electrochemistry Question with Solution | TestHub
ChemistryElectrochemistryGalvanic cells/Nernst Equation/Concentration CellsMedium2 min
ChemistryMediumsingle choice
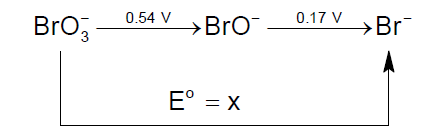
The value of "x" is
Options:
Answer:
D
Solution:
ΔG10 =?.........................(1)
ΔG20 = -4F(0.54).............(2)
ΔG30 = -2F(0.17)...............(3)
Solving the above 3 equations by
(2) + (3) = (1)
ΔG10 = -2.5VF
E = 0.417V
Stream:JEESubject:ChemistryTopic:ElectrochemistrySubtopic:Galvanic cells/Nernst Equation/Concentration Cells
⏱ 2m
Doubts & Discussion
Loading discussions...