Chemistry - Electrochemistry Question with Solution | TestHub
ChemistryElectrochemistryGalvanic cells/Nernst Equation/Concentration CellsMedium2 minPYQ_2020
ChemistryMediumstatement
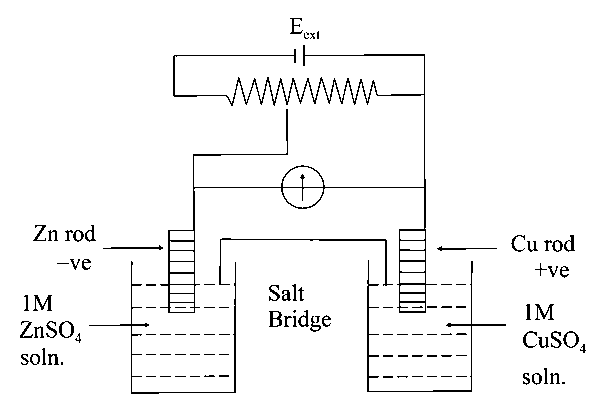
Identify the incorrect statement from the options below for the above cell:

Options:
Answer:
B
Solution:
So If no electron will flow
At cell act as electrolytic cell and electron will flow from to .
At cell act as electrochemical so dissolve and deposit.
Stream:JEESubject:ChemistryTopic:ElectrochemistrySubtopic:Galvanic cells/Nernst Equation/Concentration Cells
⏱ 2mℹ️ Source: PYQ_2020
Doubts & Discussion
Loading discussions...