Chemistry - Coordination Chemistry Question with Solution | TestHub
Total number ofcisbond angles (that is,andbonds in cis positions) present in a molecule ofcis-complex is _________
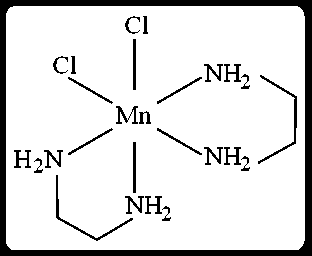
Answer:
Solution:
 The complex is cis-[Mn(en)₂(Cl)₂]. The 'en' ligand (ethylenediamine) is bidentate. In the cis isomer, the two Cl ligands are adjacent. Each 'en' ligand forms two Mn-N bonds. The two Cl ligands form two Mn-Cl bonds.
The complex is cis-[Mn(en)₂(Cl)₂]. The 'en' ligand (ethylenediamine) is bidentate. In the cis isomer, the two Cl ligands are adjacent. Each 'en' ligand forms two Mn-N bonds. The two Cl ligands form two Mn-Cl bonds.
The cis-N-Mn-Cl bond angles are formed between an Mn-N bond and an Mn-Cl bond when they are adjacent.
There are 4 Mn-N bonds (2 from each 'en' ligand) and 2 Mn-Cl bonds.
Consider one Cl ligand. It is cis to 4 N atoms (2 from each 'en'). So, 4 cis-N-Mn-Cl angles.
Since there are two Cl ligands, this gives potential cis-N-Mn-Cl angles.
However, we must avoid double counting.
Let the N atoms be N₁, N₂, N₃, N₄ and Cl atoms be Cl₁, Cl₂.
Cl₁ is cis to N₁, N₂, N₃, N₄. (4 angles)
Cl₂ is cis to N₁, N₂, N₃, N₄. (4 angles)
The total number of unique cis-N-Mn-Cl bond angles is 6.
This can be visualized by placing the two Cl ligands in cis positions (e.g., at 90 degrees to each other). Each Cl will be cis to all four N atoms.
The total number of cis-N-Mn-Cl bond angles is 6.