Chemistry - Coordination Chemistry Question with Solution | TestHub
ChemistryCoordination ChemistryIsomerismMedium2 minPYQ_2020
ChemistryMediumsingle choice
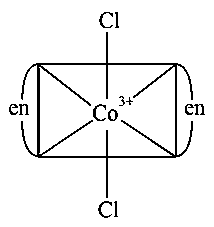
Consider the complex ions, trans-andThe correct statement regarding them is :
Options:
Answer:
D
Solution:

Have Plane of symmetry so will be optically inactive.
Trans-[Co(en)
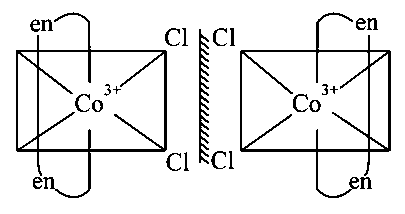
cis-[Co(en) is optically active without plane of symmetry.
Stream:JEESubject:ChemistryTopic:Coordination ChemistrySubtopic:Isomerism
⏱ 2mℹ️ Source: PYQ_2020
Doubts & Discussion
Loading discussions...