Chemistry - Coordination Chemistry Question with Solution | TestHub
ChemistryCoordination ChemistryVBT,CFT(complex compound)Easy2 minPYQ_2014
ChemistryEasysingle choice
The correct statement about the magnetic properties of [Fe(CN)6]3-and [FeF6]3-is : (Z = 26).
Options:
Answer:
A
Solution:
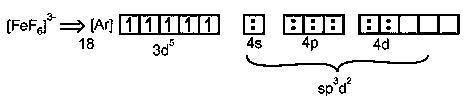
In both complexes Fe is in +3 oxidation state.
F-ion is weak field ligand so no pairing of electrons in inner 3d-orbitals & outer orbital complex. sp3d2hybridisation is formed.
have 5 unpaired electrons, so paramagnetic
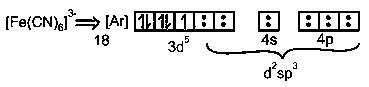
One unpaired Electron, CN-ion strong ligand field. So will pair the d-orbital electrons & form inner orbital hybridization d2sp3.
(1 unpaired e-so para magnetic)
.
Stream:JEESubject:ChemistryTopic:Coordination ChemistrySubtopic:VBT,CFT(complex compound)
⏱ 2mℹ️ Source: PYQ_2014
Doubts & Discussion
Loading discussions...