Chemistry - Complete d-block Question with Solution | TestHub
ChemistryComplete d-blockd-blockEasy2 minPYQ_2014
ChemistryEasysingle choice
A compound of vanadium chloride has spin only magnetic moment ofBM. Its formula is
Options:
Answer:
C
Solution:
Spin only Magnetic Moment
(unpaired e-)
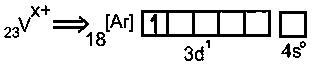
Electronic configuration of
to get 1 unpaired electronhas to lose 4 electrons to form
So, formula of chloride is
Stream:JEESubject:ChemistryTopic:Complete d-blockSubtopic:d-block
⏱ 2mℹ️ Source: PYQ_2014
Doubts & Discussion
Loading discussions...