Chemistry - Chemical Equilibrium Question with Solution | TestHub
ChemistryChemical EquilibriumProblems involving KcMedium2 minPYQ_2020
ChemistryMediumnumerical
For a reactionofofandofwere taken in avessel and allowed to react. At equilibrium, the concentration ofwas. the equilibrium constant of the reaction isThe value of x is
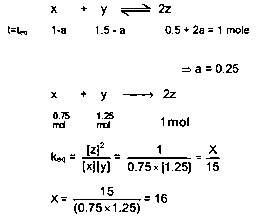
Answer:
16.00
Solution:
Since moles of are increased at equilibrium therefore reaction goes in forward direction to attain the
equilibrium.
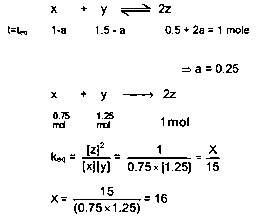
Stream:JEESubject:ChemistryTopic:Chemical EquilibriumSubtopic:Problems involving Kc
⏱ 2mℹ️ Source: PYQ_2020
Doubts & Discussion
Loading discussions...