Chemistry - Chemical Equilibrium Question with Solution | TestHub
ChemistryChemical EquilibriumEffect of temperatureHard2 minPYQ_2015
ChemistryHardsingle choice
The % yield of ammonia as a function of time in the reaction
at is given below.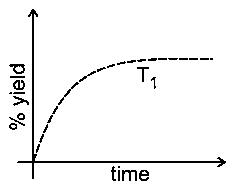
If this reaction is conducted at with , the % yield of ammonia as a function of time is represented by:

Options:
Answer:
B
Solution:
Initially on increasing temperature rate of reaction will increase, so yield will also increase with time. But at equilibrium yield at high temperature would be less than at as reaction is exothermic so the graph is

Stream:JEE_ADVSubject:ChemistryTopic:Chemical EquilibriumSubtopic:Effect of temperature
⏱ 2mℹ️ Source: PYQ_2015
Doubts & Discussion
Loading discussions...