Chemistry - Atomic Structure Question with Solution | TestHub
ChemistryAtomic Structurede-Broglie's Wave EquationMedium2 minPYQ_2024
ChemistryMediummatching list
According to the wave-particle duality of matter by de-Broglie, which of the following graph plot presents most appropriate relationship between wavelength of electronand momentum of electron
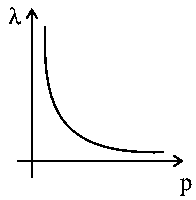
Options:
Answer:
A
Solution:
According to the de-Broglie hypothesis according to which all matters have both particle and wave nature. where plank's constant, momentum, wavelength of particle. This type of graph is called a rectangular hyperbola. As we will have a rectangular hyperbola.
(constant)
So, the plot is a rectangular hyperbola.

Stream:JEESubject:ChemistryTopic:Atomic StructureSubtopic:de-Broglie's Wave Equation
⏱ 2mℹ️ Source: PYQ_2024
Doubts & Discussion
Loading discussions...