Chemistry - Aromatic Hydrocarbon Question with Solution | TestHub
ChemistryAromatic HydrocarbonAROMATIC NUCLEPHIC SUBSTITUTION BY SNAE and SNEAEasy2 minPYQ_2023
ChemistryEasysingle choice
Arrange the following compounds in increasing order of rate of aromatic electrophilic substitution reaction.
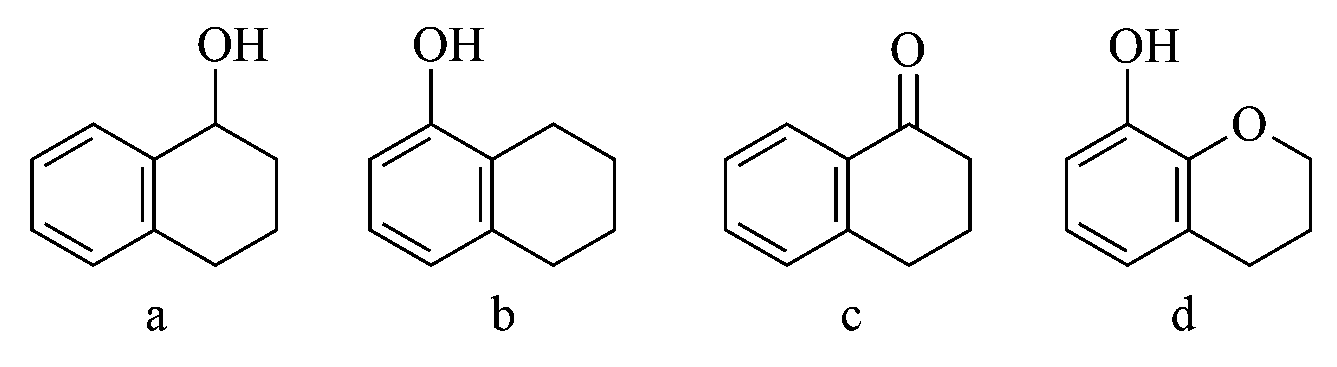

Options:
Answer:
D
Solution:
The more nucleophilic group will make ring more prone to aromatic substitution reaction. Benzene becomes more reactive towards Electrophilic Aromatic Substitution when any substituent raises its electron density.

In structure (a) group shows effect. while in (b) group shows effect. In (c) effect is shown by functional group and in both show effect.
So Correct increasing order is :
Stream:JEESubject:ChemistryTopic:Aromatic HydrocarbonSubtopic:AROMATIC NUCLEPHIC SUBSTITUTION BY SNAE and SNEA
⏱ 2mℹ️ Source: PYQ_2023
Doubts & Discussion
Loading discussions...