Physics - Heat & Thermo Question with Solution | TestHub
PhysicsHeat & ThermoDifferent processesMedium2 minPYQ_2023
PhysicsMediumsingle choice
For three low density gases pressure versus temperature graphs are plotted while keeping them at constant volume, as shown in the figure
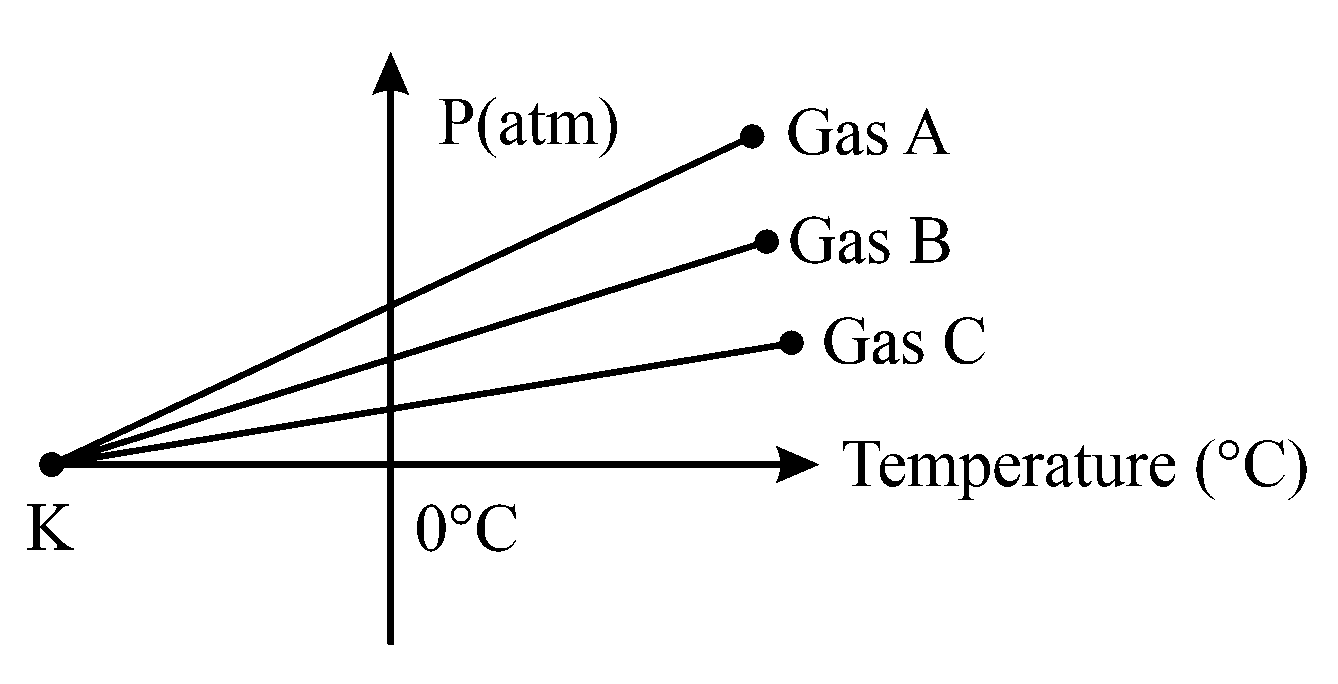
The temperature corresponding to the point is:

Options:
Answer:
A
Solution:
In case of constant volume process, i.e., isochoric process,
From ideal gas equation,
constant
Or we have,
If
Thus, when temperature goes to , all constituent particles get freezed and pressure goes to zero irrespective of the density of gas.
Hence, the temperature corresponding to the point .
Stream:JEESubject:PhysicsTopic:Heat & ThermoSubtopic:Different processes
⏱ 2mℹ️ Source: PYQ_2023
Doubts & Discussion
Loading discussions...