Physics - Heat & Thermo Question with Solution | TestHub
PhysicsHeat & ThermoFirst lawEasy2 minPYQ_2021
PhysicsEasynumerical
One mole of an ideal gas at is taken from to as shown in the given indicator diagram. The work done by the system will be _______ [Given, ] (Round off to the nearest integer)
(Round off to the nearest integer)
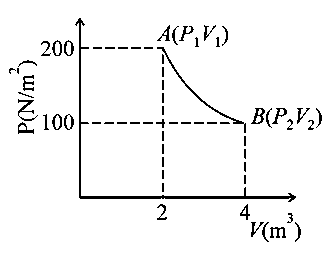

Answer:
17258.00
Solution:
Process of isothermal
Stream:JEESubject:PhysicsTopic:Heat & ThermoSubtopic:First law
⏱ 2mℹ️ Source: PYQ_2021
Doubts & Discussion
Loading discussions...