Physics - Heat & Thermo Question with Solution | TestHub
PhysicsHeat & ThermoDifferent processesHard2 minPYQ_2014
PhysicsHardsingle choice
An ideal monoatomic gas is confined in a cylinder by a spring loaded piston of cross section . Initially the gas is at and occupies a volume of and the spring is in its relaxed state as shown in figure. The gas is heated by a small heater until the piston moves out slowly by . The force constant of the spring is and the atmospheric pressure is . The cylinder and the piston are thermally insulated. The piston and the spring are massless and there is no friction between the piston and the cylinder. The final temperature of the gas will be: (Neglect the heat loss through the lead wires of the heater. The heat capacity of the heater coil is also negligible).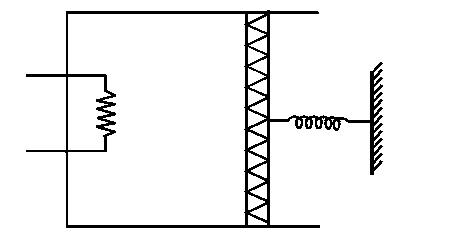

Options:
Answer:
C
Solution:
No Solution Available
Stream:JEESubject:PhysicsTopic:Heat & ThermoSubtopic:Different processes
⏱ 2mℹ️ Source: PYQ_2014
Doubts & Discussion
Loading discussions...