Physics - Heat & Thermo Question with Solution | TestHub
PhysicsHeat & ThermoDifferent processesMedium2 minPYQ_2020
PhysicsMediumnumerical
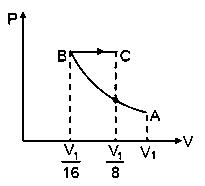
Starting at temperatureone mole of an ideal diatomic gasis first compressed adiabatically from volumetoIt is then allowed to expand isobarically to volume. If all the processes are the quasi-static then the final temperature of the gas (in) is (to the nearest integer) ___________.
Answer:
1819.00
Solution:
Now for BC process
Stream:JEESubject:PhysicsTopic:Heat & ThermoSubtopic:Different processes
⏱ 2mℹ️ Source: PYQ_2020
Doubts & Discussion
Loading discussions...