Physics - Heat & Thermo Question with Solution | TestHub
PhysicsHeat & ThermoDifferent processesEasy2 minPYQ_2015
PhysicsEasysingle choice
An ideal gas goes through a reversible cyclehas the V - T diagram shown below. Processare adiabatic.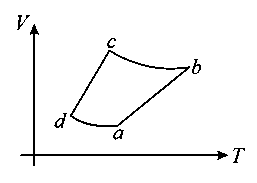
The corresponding P - V diagram for the process is (all figures are schematic and not drawn to scale) :

Options:
Answer:
A
Solution:
Is an adiabatic process
as T increase V decreases at non-uniform rate
In processP = constant as
In processconstant s
But since slope of V - T graph
since slope of ab < slope of cd
Also in adiabatic processas T is increasing V in decreasing
P is increasing, so P - V diagram is as below
Stream:JEESubject:PhysicsTopic:Heat & ThermoSubtopic:Different processes
⏱ 2mℹ️ Source: PYQ_2015
Doubts & Discussion
Loading discussions...