Chemistry - Thermodynamics - I Question with Solution | TestHub
ChemistryThermodynamics - IIsothermal Process/Isobaric/IsochoricMedium2 minPYQ
ChemistryMediumsingle choice
Arrange the following in order of magnitude of work done by the system / on the system at constant temperature :
(a) for expansion in infinite stage.
(b) for expansion in single stage.
(c) for compression in infinite stage.
(d) for compression in single stage.
Choose the correct answer from the options given below:
Options:
Answer:
B
Solution:
For isothermal process
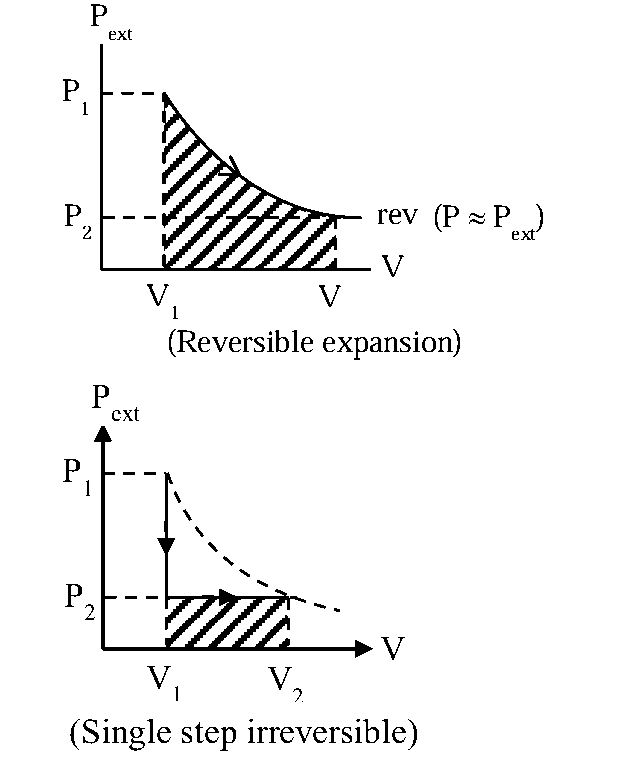

We can compare work by area of PV graph
Stream:JEESubject:ChemistryTopic:Thermodynamics - ISubtopic:Isothermal Process/Isobaric/Isochoric
⏱ 2mℹ️ Source: PYQ
Doubts & Discussion
Loading discussions...