Chemistry - Salt Analysis Question with Solution | TestHub
ChemistrySalt AnalysisANION miscellaneous/mixedMedium2 minPYQ_2022
ChemistryMediumstatement
A white precipitate was formed whenwas added to water extract of an inorganic salt. Further, a gaswith characteristic odour was released when the formed white precipitate was dissolved in dilute HCI. The anion present in the inorganic salt is:
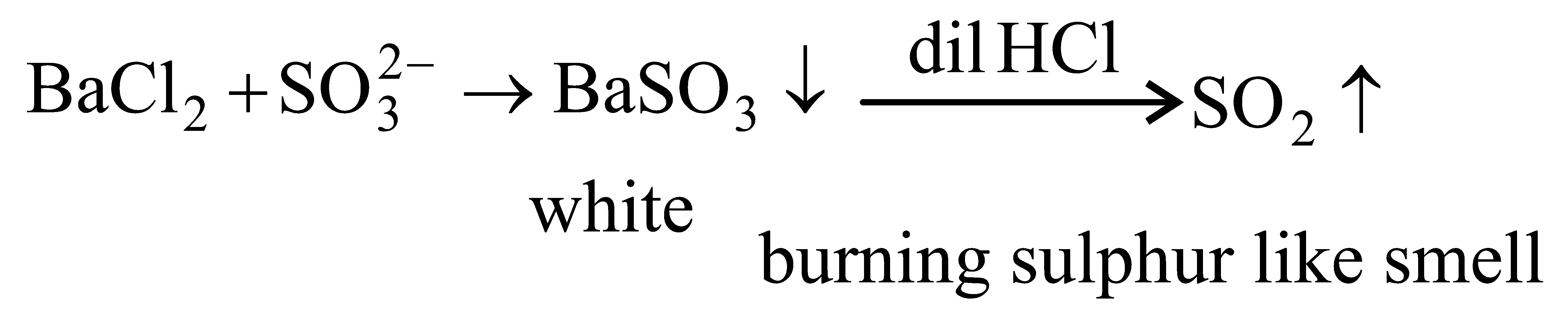
Options:
Answer:
B
Solution:
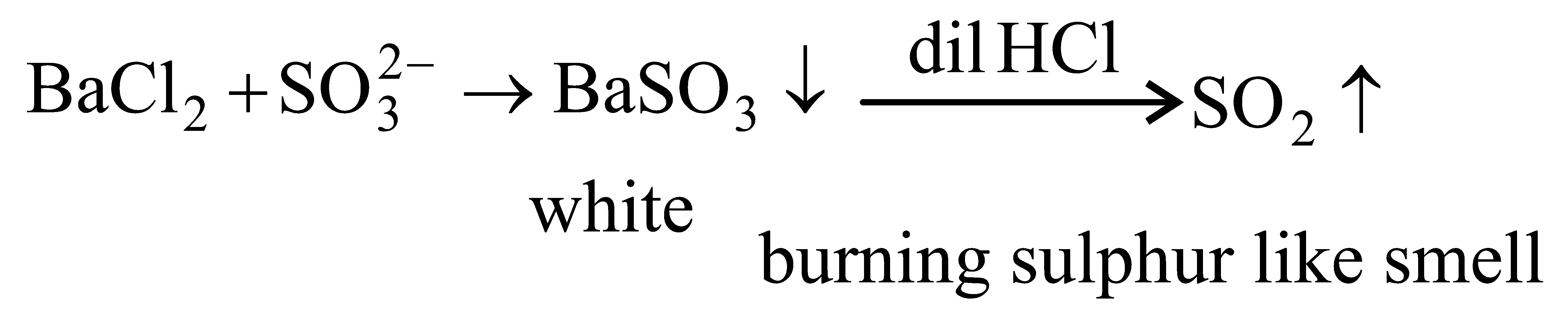
A white precipitate was formed when when added to water extract of salt. which is an inorganic salt. is formed. Further, a gas with burning sulphur like odour will be released when the is dissolved in dilute HCI.
Stream:JEESubject:ChemistryTopic:Salt AnalysisSubtopic:ANION miscellaneous/mixed
⏱ 2mℹ️ Source: PYQ_2022
Doubts & Discussion
Loading discussions...