Chemistry - Salt Analysis Question with Solution | TestHub
ChemistrySalt AnalysisCATION miscellaneous/mixedMedium2 minPYQ_2017
ChemistryMediummultiple choice
Addition of excess aqueous ammonia to a pink coloured aqueous solution ofandgives an octahedral complex Y in the presence of air. In aqueous solution, complex Y behaves as 1 : 3 electrolyte. The reaction of X with excess HCl at room temperature results in the formation of a blue coloured complex Z. The calculated spin only magnetic moment of X and Z is 3.87 B.M., whereas it is zero for complex Y.
Among the following options, which statement(s) is(are) correct ?
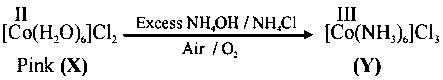
Options:(select one or more)
Answer:
A, B, C
Solution:
Cobalt salts are pink in anhydrous & blue in hydrated form
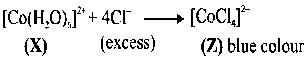
(i) Hybridisation of (Y) is as is a strong field ligand
(ii) have hybridisation as is a weak field ligand
(iii) 
(iv) 
Stream:JEESubject:ChemistryTopic:Salt AnalysisSubtopic:CATION miscellaneous/mixed
⏱ 2mℹ️ Source: PYQ_2017
Doubts & Discussion
Loading discussions...