Chemistry - PRACTICAL ORGANIC CHEMISTRY Question with Solution | TestHub
ChemistryPRACTICAL ORGANIC CHEMISTRYDIFFERENTIATION, SEPARATION AND PURIFICATION of ORGANIC COMPOUNDSEasy2 minPYQ_2023
ChemistryEasynumerical
Three organic compounds and were allowed to run in thin layer chromatography using hexane and gave the following result (see figure). The value of the most polar compound is _____
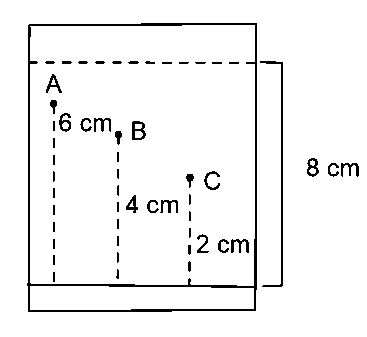

Answer:
25.00
Solution:
In thin layer chromatography using hexane, the least polar compound will rise to maximum height and most polar compound will rise to minimum height.
The retardation factor of each spot can be determined by dividing the distance that a particular compound has travelled by the distance between solvent front and initial spotting site. This parameter is dependent on the thin layer chromatographic plate and the solvent.
value for most polar compound
Stream:JEESubject:ChemistryTopic:PRACTICAL ORGANIC CHEMISTRYSubtopic:DIFFERENTIATION, SEPARATION AND PURIFICATION of ORGANIC COMPOUNDS
⏱ 2mℹ️ Source: PYQ_2023
Doubts & Discussion
Loading discussions...