Chemistry - Ionic Equilibrium Question with Solution | TestHub
ChemistryIonic EquilibriumTitration Based ProblemsMedium2 minPYQ_2019
ChemistryMediumstatement
In an acid-base titration,solution was added to thesolution of unknown strength. Which of the following correctly shown the change ofof the titration mixture in this experiment?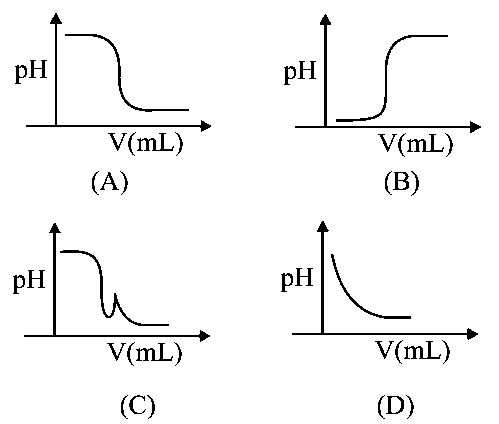

Options:
Answer:
A
Solution:
For titration of strong base with strong acidchanges sharply near equivalence point and at equivalent pointsharply decreases because strong base & strong acid dissociate completely.
at equivalent pointand at endpoint solution will be neutral.
Stream:JEESubject:ChemistryTopic:Ionic EquilibriumSubtopic:Titration Based Problems
⏱ 2mℹ️ Source: PYQ_2019
Doubts & Discussion
Loading discussions...