Chemistry - Hydrocarbon Question with Solution | TestHub
ChemistryHydrocarbonGeneralMedium2 minPYQ_2025
ChemistryMediumsingle choice
Following are the four molecules "P", "Q", "R" and "S".
Which one among the four molecules will react with at the fastest rate?


Options:
Answer:
C
Solution:
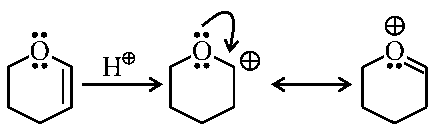
Addition of to alkene follows electrophilic addition mechanism. In the rate determining step a carbocation intermediate is formed. Among , R \& S compound Q will form most stable carbocation intermediate since it is resonance stabilized.
Stream:JEESubject:ChemistryTopic:HydrocarbonSubtopic:General
⏱ 2mℹ️ Source: PYQ_2025
Doubts & Discussion
Loading discussions...