Chemistry - Halogen Derivative Question with Solution | TestHub
Given below are two statements : one is labelled as Assertion and the other is labelled as Reason
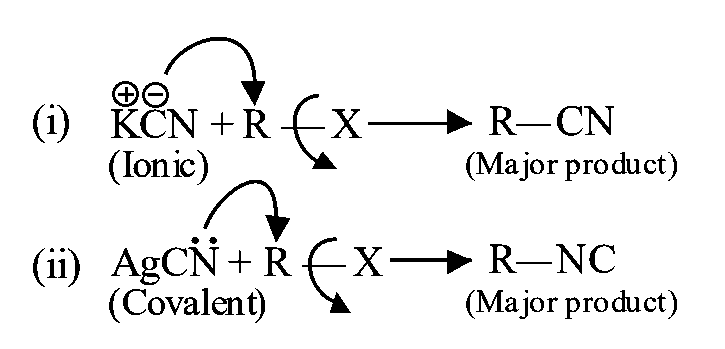
Assertion Haloalkanes react with to form alkyl cyanides as a main product while with form isocyanide as the main product.
Reason and both are highly ionic compounds.
In the light of the above statement, choose the most appropriate answer from the options given below.
Options:
Answer:
Solution:
is ionic in nature and provides cyanide ions in solution. Although both carbon and nitrogen atoms are in a position to donate electron pairs, the attack takes place mainly
through carbon atom and not through nitrogen atom since bond is more stable than bond.
Involved reaction is shown below:
Reaction of alkyl halide with
is mainly covalent in nature. Here only nitrogen is free to donate electron pairs,hence it forms isocyanide as the main product.
Involved reaction is shown below:
Hence, here isocyanide is formed as major product.

is mainly covalent in nature and nitrogen is available for attack, so alkyl isocyanide is formed as main product.
Cyanide ion is an ambidentate ligand (nucleophile or lewis base). It means that donation of a pair of electrons can be through either carbon or nitrogen, so it has two nucleophilic centres. is predominantly ionic and is predominantly covalent.