Chemistry - GOC Question with Solution | TestHub
ChemistryGOCRESONANCE AND MESOMERIC EFFECTMedium2 minPYQ_2023
ChemistryMediumsingle choice
The correct order for acidity of the following hydroxyl compound is
(A)
(B)
(C) 
(D) 
(E) 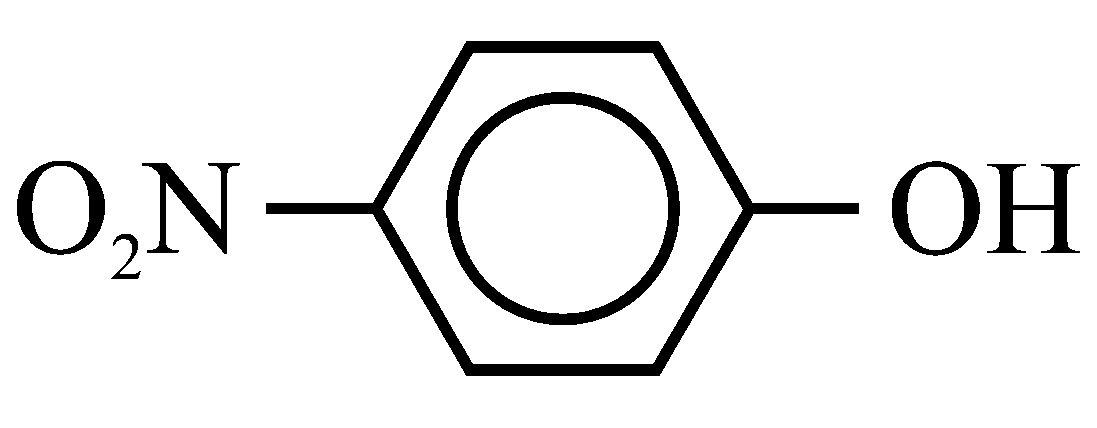
Choose the correct answer from the options given below:

Options:
Answer:
D
Solution:
The compounds A & B are alcohols, and rest of the compounds are phenols. Phenols are more acidic than alcohols. Among the A &B, methyl alcohol is more acidic than tertiary butyl alcohol as alkyl groups decreases the acidic nature by electron releasing tendency. Among the phenols, the phenol with electron withdrawing group is more acidic. (Electron withdrawing group) group will increase the acidity, (Electron releasing group) group at para position will decrease the acidity.
So, Acidic strength E>C>D>A>B
Stream:JEESubject:ChemistryTopic:GOCSubtopic:RESONANCE AND MESOMERIC EFFECT
⏱ 2mℹ️ Source: PYQ_2023
Doubts & Discussion
Loading discussions...