Chemistry - GOC Question with Solution | TestHub
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
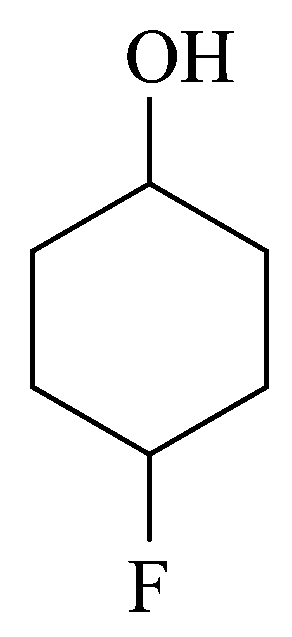
Assertion A : Order of acidic nature of the following compounds is .
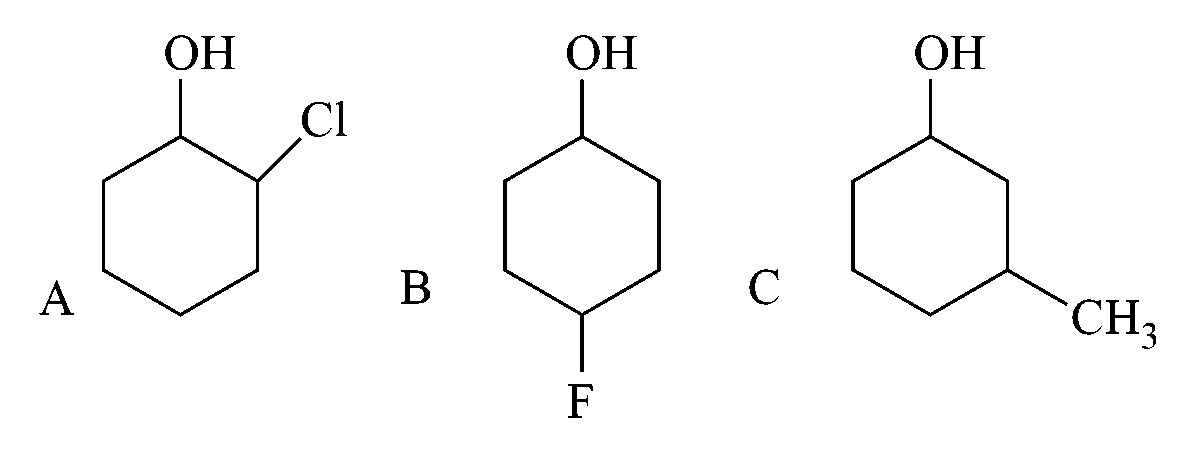
Reason R: Fluoro is a stronger electron withdrawing group than Chloro group.
In the light of the above statements, choose the correct answer from the options given below :

Options:
Answer:
Solution:
The electron withdrawing groups decreases the acidic nature and The electron releasing groups increases the acidic nature.
The are electron withdrawing while is electron loosing hence acidic strength will be

On further differentiation the effect depends on most importantly over distance hence.

The has higher effect as compared to Cl hence reason is correct statement but the effect depends more on distance as compared to power hence  is stronger acid than
is stronger acid than