Chemistry - GOC Question with Solution | TestHub
ChemistryGOCRESONANCE AND MESOMERIC EFFECTMedium2 minPYQ_2021
ChemistryMediumsingle choice
Compound(s) which will liberate carbon dioxide with sodium bicarbonate solution is/are :


Options:
Answer:
B
Solution:
Compounds which are more acidic then , gives gas on reaction with . Compound i.e. Benzoic acid and compound i.e. picric acid both are more acidic than .
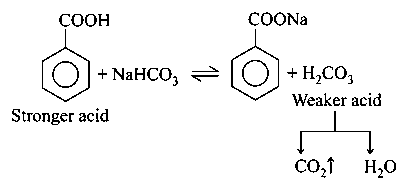
equilibrium favours forward direction and is liberated.

Equilibrium favours backward direction and is not liberated.
Stream:JEESubject:ChemistryTopic:GOCSubtopic:RESONANCE AND MESOMERIC EFFECT
⏱ 2mℹ️ Source: PYQ_2021
Doubts & Discussion
Loading discussions...