Chemistry - GOC Question with Solution | TestHub
ChemistryGOCAromaticityMedium2 minPYQ_2019
ChemistryMediumsingle choice
Among the following four aromatic compounds, which one will have the lowest melting point?

Options:
Answer:
C
Solution:
Melting point ∝ force of attraction. In  and
and

hydrogen bonding takes place. Hence, the maximum force of attraction is present among molecules. The compound 
is polar in nature due to the carbonyl group and thus, will have moderately strong dipole-dipole interaction. This will give them a high melting point. In
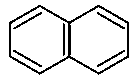
only van der Waal's force of attraction (weak force) is present. Hence, it will have the lowest melting point.
Stream:JEESubject:ChemistryTopic:GOCSubtopic:Aromaticity
⏱ 2mℹ️ Source: PYQ_2019
Doubts & Discussion
Loading discussions...