Chemistry - GOC Question with Solution | TestHub
ChemistryGOCAromaticityHard2 minPYQ_2019
ChemistryHardsingle choice
The increasing order of the values of the following compounds is: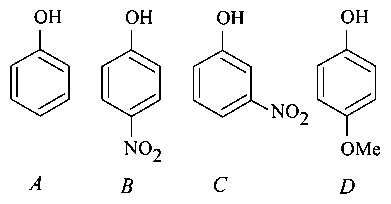

Options:
Answer:
A
Solution:
& vice versa
Acidic character
Acidic character α stability of anion (Conjugate base)
Conjugate base are
Stability of conjugate base
is more dominating than
at para so exerts or & >
So it is highly stable anion in
at meta exerts only effect
No withdrawing or electron releasing group.
has but dominates
So acidic strength will be
Therefore values will be in the order
Stream:JEESubject:ChemistryTopic:GOCSubtopic:Aromaticity
⏱ 2mℹ️ Source: PYQ_2019
Doubts & Discussion
Loading discussions...