Chemistry - GOC Question with Solution | TestHub
ChemistryGOCRESONANCE AND MESOMERIC EFFECTEasy2 minPYQ_2014
ChemistryEasysingle choice
Considering the basic strength of amines in an aqueous solution, which one has the smallestvalue?
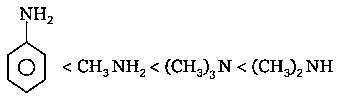
Options:
Answer:
A
Solution:
Since in aromatic amines the lone pair undergoes delocalisation, aliphatic amines are more basic than aromatic amines.
In an aliphatic amine, due to the effect of the order will be .
In an aqueous solution, bonding takes place. So, the order will be .
However, the resultant basic order is due to a mixture of both the effects( and hydrogen bond). So, the order is .
So, the final basic order is 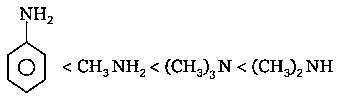
Stream:JEESubject:ChemistryTopic:GOCSubtopic:RESONANCE AND MESOMERIC EFFECT
⏱ 2mℹ️ Source: PYQ_2014
Doubts & Discussion
Loading discussions...