Chemistry - Electrochemistry Question with Solution | TestHub
ChemistryElectrochemistryFaraday's Law of Electrolysis/Electrolytic CellsMedium2 minPYQ_2016
ChemistryMediumsingle choice
Oxidation of succinate ion produces ethylene and carbon dioxide gases. On passingFaraday electricity through an aqueous solution of potassium succinate, what is the total volume of gases (at both cathode and anode) at STP (and)?
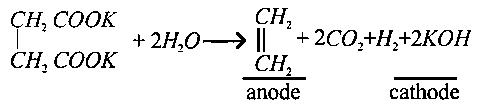
Options:
Answer:
A
Solution:
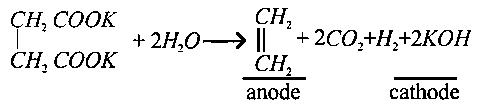
Ethene, are gases.
So total moles of gas
By Farady mole of gas produced
So, by Farady mole gas produced
Stream:JEESubject:ChemistryTopic:ElectrochemistrySubtopic:Faraday's Law of Electrolysis/Electrolytic Cells
⏱ 2mℹ️ Source: PYQ_2016
Doubts & Discussion
Loading discussions...