Chemistry - Coordination Chemistry Question with Solution | TestHub
ChemistryCoordination ChemistryIsomerismHard2 minPYQ_2023
ChemistryHardsingle choice
Chiral complex from the following is :
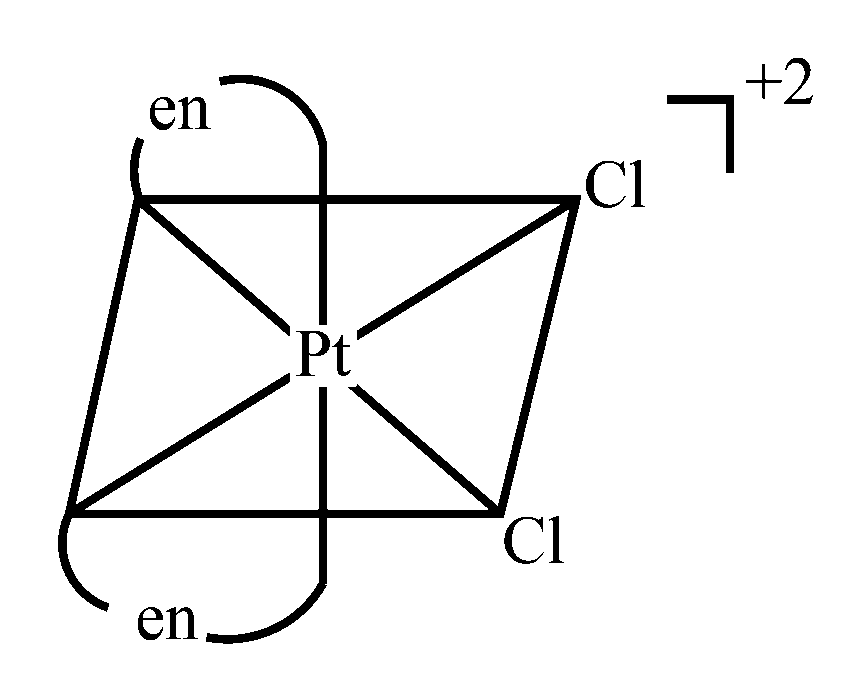
Here en = ethylene diamine
Options:
Answer:
A
Solution:
Here en = ethylene diamine

[PtCl2(en)2]2+ forms cis and trans-isomers. Only the cis-isomers has no element of symmetry. So it is optically active. Trans - [PtCl2(en)2]2+ form is optically inactive. Square planar complexes doesn't exhibit optical isomerism.
Stream:JEESubject:ChemistryTopic:Coordination ChemistrySubtopic:Isomerism
⏱ 2mℹ️ Source: PYQ_2023
Doubts & Discussion
Loading discussions...