Chemistry - Coordination Chemistry Question with Solution | TestHub
ChemistryCoordination ChemistryVBT,CFT(complex compound)Medium2 minPYQ_2023
ChemistryMediummatching list
Match List-I with List-II
| LIST-I Coordination Complex | LIST-II Number of unpaired electrons | ||
| A. | I. | 0 | |
| B. | II. | 3 | |
| C. | III. | 2 | |
| D. | IV. | 4 |
Choose the correct answer from the options given below:

Options:
Answer:
A
Solution:
ion, oxidation state of is +3 and its valence shell electronic configuration is . There are unpaired electrons in orbital.
(A).
No. of unpaired electrons
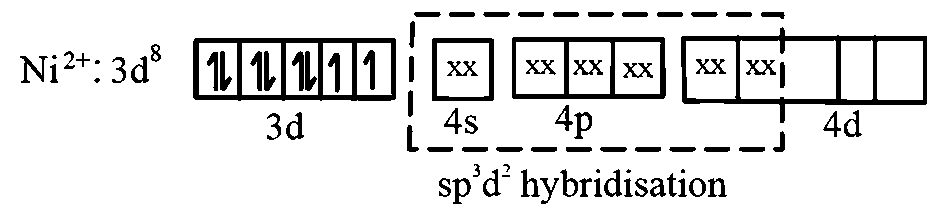
ion, oxidation state of is +2 and its valence shell electronic configuration is . There are 4 unpaired electrons in 3d orbital. So, you can say the hybridisation here would be .
(B).
No. of unpaired electronsin this oxidation state of central metal atom is and it has no unpaired electrons.
(C).
No. of unpaired electronsthe oxidation state of is and it has two unpaired electrons.
(D)
No. of unpaired electrons
So the correct option among the given is .
Stream:JEESubject:ChemistryTopic:Coordination ChemistrySubtopic:VBT,CFT(complex compound)
⏱ 2mℹ️ Source: PYQ_2023
Doubts & Discussion
Loading discussions...