Chemistry - Coordination Chemistry Question with Solution | TestHub
ChemistryCoordination ChemistryVBT,CFT(complex compound)Hard2 minPYQ_2022
ChemistryHardsingle choice
Arrange the following coordination compounds in the increasing order of magnetic moments. (Atomic numbers: )
A.
B.
C. (high spin)
D.
Choose the correct answer from the options given below

Options:
Answer:
B
Solution:
Magnetic moment is directly proportional to the number of unpaired in the central metal atom of a complex.
It is calculated by the formula,
(A)
(B)

(C)
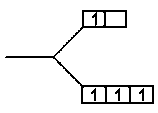
(D)
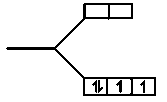
Stream:JEESubject:ChemistryTopic:Coordination ChemistrySubtopic:VBT,CFT(complex compound)
⏱ 2mℹ️ Source: PYQ_2022
Doubts & Discussion
Loading discussions...