Chemistry - Coordination Chemistry Question with Solution | TestHub
ChemistryCoordination ChemistryVBT,CFT(complex compound)Hard2 minPYQ_2019
ChemistryHardsingle choice
The complex ion that will lose its crystal field stabilization energy upon oxidation of metal tostate is:

Options:
Answer:
B
Solution:
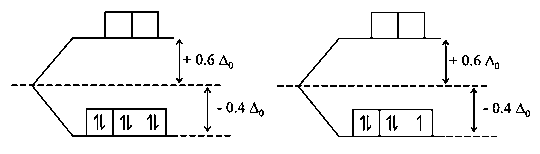
In all other cases, the electron is removed from orbital and is hence, this will lead to decrease in pairing energy.
Stream:JEESubject:ChemistryTopic:Coordination ChemistrySubtopic:VBT,CFT(complex compound)
⏱ 2mℹ️ Source: PYQ_2019
Doubts & Discussion
Loading discussions...