Chemistry - CHEMICAL BONDING Question with Solution | TestHub
ChemistryCHEMICAL BONDINGVSEPR, Bond Angle, Bond Length, Bond EnergyMedium2 minPYQ_2022
ChemistryMediumsingle choice
In the structure of, the lone pair of electrons onis in.
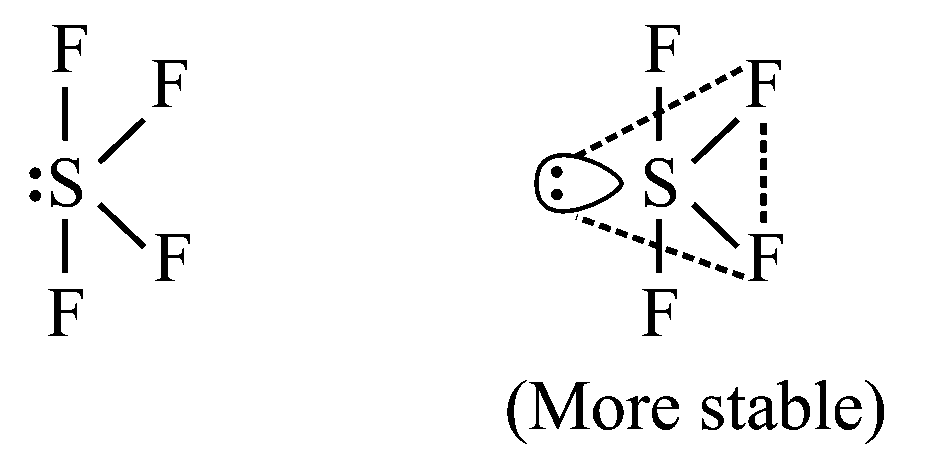
Options:
Answer:
A
Solution:
In , The lone pair is in an equatorial position, and there are two lp—bp repulsions. Hence, the following arrangement shown below is more stable.
The shape shown in the following figure is described as a distorted tetrahedron, a folded square or a see-saw.
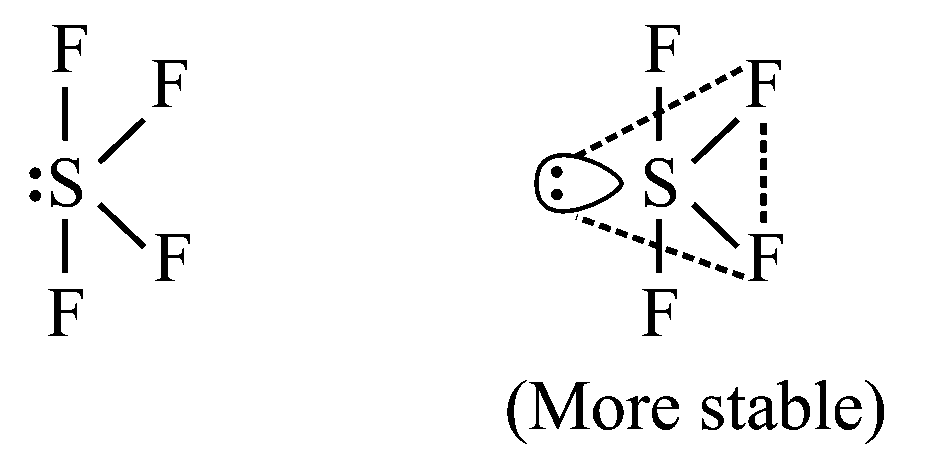
Stream:JEESubject:ChemistryTopic:CHEMICAL BONDINGSubtopic:VSEPR, Bond Angle, Bond Length, Bond Energy
⏱ 2mℹ️ Source: PYQ_2022
Doubts & Discussion
Loading discussions...