Chemistry - CHEMICAL BONDING Question with Solution | TestHub
ChemistryCHEMICAL BONDINGVSEPR, Bond Angle, Bond Length, Bond EnergyMedium2 minPYQ_2021
ChemistryMediumsingle choice
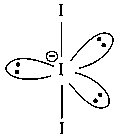
The correct shape andbond angles respectively inion are:
Options:
Answer:
D
Solution:
The central iodine atom undergoes hybridization and has 3 lone pairs of electrons and 2 bond pairs of electrons.
Thus, has trigonal bipyramidal geometry and linear shape .

Shape : Linear, I-I-I Bond angle
Stream:JEESubject:ChemistryTopic:CHEMICAL BONDINGSubtopic:VSEPR, Bond Angle, Bond Length, Bond Energy
⏱ 2mℹ️ Source: PYQ_2021
Doubts & Discussion
Loading discussions...