Chemistry - CHEMICAL BONDING Question with Solution | TestHub
ChemistryCHEMICAL BONDINGVSEPR, Bond Angle, Bond Length, Bond EnergyEasy2 minPYQ_2020
ChemistryEasysingle choice
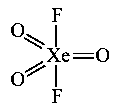
The shape/structure ofand, respectively are :

Options:
Answer:
A
Solution:
(i)
St. No. so hybridisation is and structure is pentagonal planar.

(ii) St. No.
so hybridisation is and structure is trigonal bipyramidal.
Stream:JEESubject:ChemistryTopic:CHEMICAL BONDINGSubtopic:VSEPR, Bond Angle, Bond Length, Bond Energy
⏱ 2mℹ️ Source: PYQ_2020
Doubts & Discussion
Loading discussions...