Chemistry - CHEMICAL BONDING Question with Solution | TestHub
ChemistryCHEMICAL BONDINGDipole MomentEasy2 minPYQ_2014
ChemistryEasysingle choice
For which of the following molecule significant?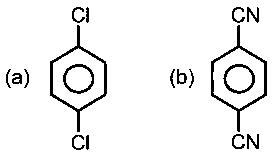
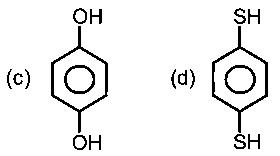

Options:
Answer:
D
Solution:
In (C) and (D) the oxygen and sulphur are sp3 hybridized, with 2 lone pairs. Thus the molecule is 3D, and has a dipole moment.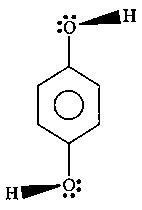
(Net dipole outside the plane at paper)
Stream:JEESubject:ChemistryTopic:CHEMICAL BONDINGSubtopic:Dipole Moment
⏱ 2mℹ️ Source: PYQ_2014
Doubts & Discussion
Loading discussions...