Chemistry - CHEMICAL BONDING Question with Solution | TestHub
ChemistryCHEMICAL BONDINGWeak ForcesMedium2 minPYQ_2014
ChemistryMediummultiple choice
Hydrogen bonding plays a central role in the following phenomena:
Options:(select one or more)
Answer:
A, B, D
Solution:
A] Ice has cage-like structure in which each water molecule is surrounded by four other water molecules tetrahedral through hydrogen boding, due to this density of ice is less than water and it floats in water.
B]
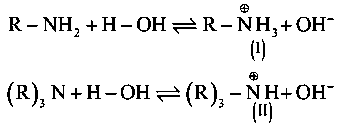
The cation (I) more stabilized through hydrogen boding than cation II . So, is better base than in aqueous solution.
C] HCOOH is stronger acid than due to inductive effect and not due to hydrogen bonding.
D] Acetic acid dimerizes in benzene through intermolecular hydrogen bonding.

Stream:JEE_ADVSubject:ChemistryTopic:CHEMICAL BONDINGSubtopic:Weak Forces
⏱ 2mℹ️ Source: PYQ_2014
Doubts & Discussion
Loading discussions...