Chemistry - Atomic Structure Question with Solution | TestHub
ChemistryAtomic StructureSchrödinger's EquationMedium2 minPYQ_2016
ChemistryMediumstatement
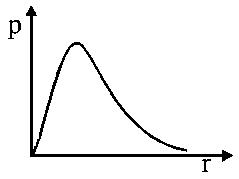
is the probability of finding theelectron of the hydrogen atom in a spherical shell of infinitesimal thickness,, at a distance,, from the nucleus. The volume of this shell is. The qualitative sketch of the dependence ofonis:
Options:
Answer:
C
Solution:
For, the radial part of the wave function is:
Probability of finding anin a spherical shell of thickness,at a distancefrom the nucleus:
So,is zero atand.
So, the plot of radial probability function,V/sis as shown above.
Stream:JEE_ADVSubject:ChemistryTopic:Atomic StructureSubtopic:Schrödinger's Equation
⏱ 2mℹ️ Source: PYQ_2016
Doubts & Discussion
Loading discussions...