Chemistry - Atomic Structure Question with Solution | TestHub
ChemistryAtomic StructureBohr's Atomic ModelEasy2 minPYQ_2006
ChemistryEasymatching list
According to Bohr's theory Total energy, Kinetic energy, Potential energy, Radius of th orbit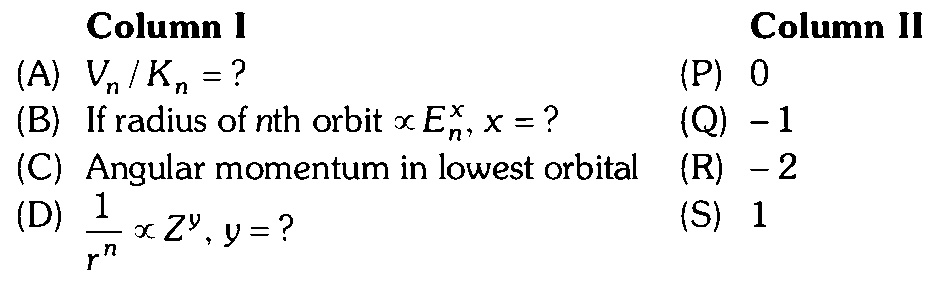
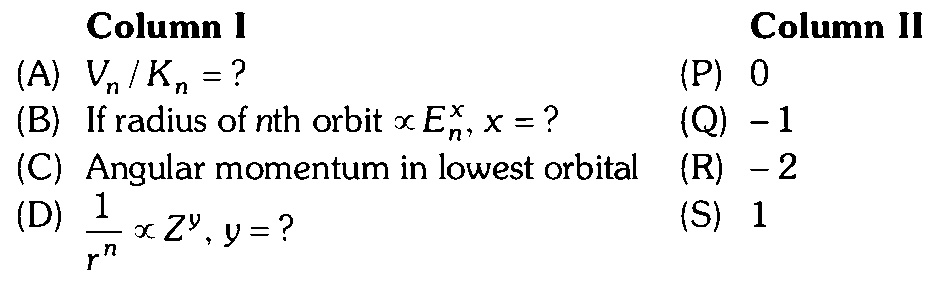
Options:
Answer:
C
Solution:
According to Bohr's theory, Total energy is Kinetic energy Potential energy Radius of th orbit (A) Hence, match with . (B) or Radius of th orbit Hence, (B) match with (Q). (C) Angular momentum For the lower orbit and Hence, angular momentum of lowest orbit (C) match with (P) (D) as Hence, (D) match with (S).
Stream:JEE_ADVSubject:ChemistryTopic:Atomic StructureSubtopic:Bohr's Atomic Model
⏱ 2mℹ️ Source: PYQ_2006
Doubts & Discussion
Loading discussions...